Turnkey Solution for Medical Disposable Urine Collection Bag Manufacturing
Overview

A Urine Bag Production Line is a complete turnkey manufacturing system designed for the mass production of disposable medical urine collection bags, including bedside urine bags and leg urine bags.
Unlike a single machine, a full urine bag production line integrates material handling, RF welding, tube assembly, leak testing, sterilization, and packaging, while complying with international medical standards such as ISO 13485 and GMP.
This production line is widely used by:
- Medical disposable manufacturers
- New investors entering medical consumables
- Hospitals and healthcare supply projects
- Government and NGO medical programs
At productionline.group, we provide end-to-end urine bag production line solutions, from factory layout design to equipment configuration and production ramp-up.
Types of Urine Bags Covered by the Line
A well-designed urine bag production line can manufacture multiple product types:
- Bedside Urine Bags
- Capacity: 1000 ml / 1500 ml / 2000 ml
- Used in hospitals and clinics
- Leg Urine Bags
- Compact design for patient mobility
- Usually supplied with straps and anti-reflux valves
- Material Options
- Medical-grade PVC
- DEHP-free PVC (EU-compliant)
The line configuration can be adjusted according to product size, thickness, and target market regulations.
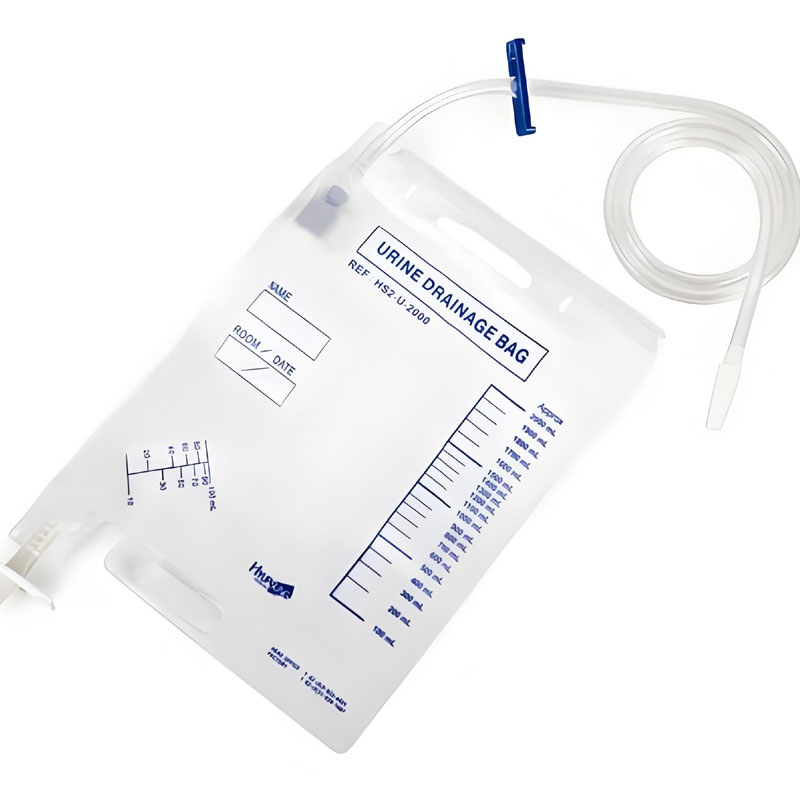
Complete Urine Bag Production Line Configuration
A standard turnkey urine bag production line includes the following sections:
1. Film Preparation / Film Supply
- Medical-grade PVC film feeding
- Thickness control and tension adjustment
2. RF Welding & Bag Forming Machine
- High-frequency RF welding for strong, uniform seams
- Automatic bag forming and edge sealing
- Stable welding quality suitable for long-term mass production
3. Tube Assembly & Valve Insertion
- Automatic tube cutting and insertion
- Anti-reflux valve assembly
- Leak-proof joint welding
4. Leak Testing System
- 100% leakage inspection
- Air or water pressure testing
- Ensures product safety before sterilization
5. EO / Gamma Sterilization (Optional)
- EO sterilization is the most common choice
- Compatible with medical regulatory requirements
- Gamma sterilization optional depending on target market
6. Packing & Labeling Line
- Medical pouch packing
- Label printing and traceability coding
- Carton packing for shipment
Each module can be customized based on production capacity and automation level.

Production Capacity Reference
Typical output ranges (for reference):
- Semi-automatic line:
800 – 1,500 pcs/hour - Fully automatic line:
2,000 – 4,000 pcs/hour
Actual capacity depends on:
- Bag size
- Welding complexity
- Automation level
- Operator experience
Cleanroom & Factory Requirements
Urine bag production requires a controlled environment:
- Cleanroom Class:
- ISO Class 8 (recommended)
- ISO Class 7 for high-end markets
- Utility Requirements:
- Stable electrical power supply
- Compressed air
- EO sterilization room (if applicable)
- Compliance Standards:
- ISO 13485
- GMP
- Local medical device registration regulations
We assist customers in factory layout planning, cleanroom zoning, and production flow optimization.

Turnkey Project Scope
Our urine bag production line solution is not limited to equipment supply. It covers:
- Production process design
- Equipment selection & integration
- Cleanroom layout guidance
- Installation & commissioning
- Operator training
- Production trial & quality validation
This turnkey approach helps investors reduce startup risks and shorten the time to market.
Investment & ROI Considerations
Key factors affecting project investment:
- Automation level
- Production capacity
- Sterilization method
- Local labor cost
- Regulatory compliance cost
A properly designed urine bag production line can achieve:
- Stable product quality
- Predictable operating cost
- Fast return on investment for emerging markets
We provide capacity-based configuration instead of over-sizing equipment.
Frequently Asked Questions (FAQ)
Q1: Can one production line produce different sizes of urine bags?
Yes. With mold and parameter adjustments, one line can cover multiple bag sizes.
Q2: Is EO sterilization mandatory?
Most medical markets require sterilization. EO is the most common and flexible solution.
Q3: What materials are supported?
Medical-grade PVC and DEHP-free PVC are both supported.
Q4: How many operators are needed?
Fully automatic lines typically require 2–4 operators per shift.
Q5: What certifications are required?
ISO 13485 and GMP are recommended. Local medical registration depends on the destination market.
Q6: Can the line be expanded later?
Yes. The system can be designed with future capacity expansion in mind.
Q7: What is the typical delivery time?
Normally 60–90 days depending on configuration.
Q8: Do you provide installation and training?
Yes. Remote or on-site support can be arranged.
Why Choose a Turnkey Urine Bag Production Line?
Choosing a complete production line instead of individual machines ensures:
- Consistent product quality
- Higher production efficiency
- Easier regulatory compliance
- Lower long-term operating risk
A urine bag production line is not just equipment—it is a medical manufacturing system.
Contact Us for a Customized Solution
If you are planning to invest in a urine bag manufacturing project, we can help you design a production line tailored to your target market, budget, and compliance requirements.
📩 Contact us to receive:
- Line configuration proposal
- Factory layout suggestion
- Investment estimation